Based on a true story. Details have been changed and obfuscated for the sake of privacy.
When I experience a challenging case (with regards to medical management), I like to learn from wisdom of #medtwitter. I’ll share my thought process throughout 24 hours of this case, as well as important insight from clinicians that responded to poll.
A middle aged woman presents to your ICU as referral from an outlying hospital due to refractory hypoxemia. She had been treated in a progressive care unit for several days on alternating HFNC and NIV, finally placed on invasive mechanical ventilation the day of transfer. Prone position ventilation was attempted but saturations remained in the 80s on 100% oxygen. In the process, she developed two pressor shock. She reportedly had an echocardiogram while hospitalized with normal LV and RV function, but at least moderately severe mitral regurgitation.
She arrived to my ICU on 8 ml/kg tidal volume, 12 of PEEP, with a driving pressure of 22 (i.e. plateau pressure 34). Initial ABG: 7.32/PaCO2 50/PaO2 70 on 100% O2.
Current pressor doses are norepinephrine 0.5 mcg/kg/min and vasopressin 0.03 units/min). She is sedated and receiving neuromuscular blockade. Fingers and toes are frankly purple; capillary refill time is 8 seconds. An intermittently dampened radial arterial line is in place, as well as a dual lumen PICC which is occupied with essential medications. Inhaled epoprostenol is started.
This patient has clearly severe circulatory shock and gas exchange impairment. Her risk of dying is extremely high. What should we do? Giving only the limited data below, I posed a poll and got the following responses:

Starting simple
Clearly the current ventilatory parameters are unsafe and can be optimized. Tidal volume is dropped to 6 ml/kg, driving pressure improves to 18. Uptitration of PEEP (high as 16) results in worsening hemodynamics, so a PEEP of 8 is trialed. Driving pressure falls further, and FiO2 is able to be weaned to 75%. Shock has not significantly changed from prior.
Bedside ultrasound is notable for the following:
- Lung US shows mixed A > B line pattern in the left lung. There is clear hepatization of the bottom 2/3 of the right lung
- Transthoracic echo images are quite limited. We were able to see from the subcostal view well enough to discern the RV was not grossly enlarged, the LV looked moderately enlarged and perhaps with moderate systolic dysfunction but it was difficult to say from the view. We were able to perform a bedside bubble study to confirm absence of significant right to left shunt. We were unable to obtain views to estimate cardiac output via LVOT VTI.
We weren’t sure how old the PICC line was, and it was insufficient for access, so a new line had to be placed. We clearly need better assessment of the shock state, and to a lesser extent, the respiratory failure. What’s next?
Check your inventory
- What was available to me: central venous catheter, pulmonary artery catheter, non-calibrated CO devices
- What wasn’t available that would have been useful: calibrated CO devices (e.g. PiCCO), transesophageal echo
What makes sense?
The ESICM consensus on circulatory shock and hemodynamic monitoring makes the following recommendations which are useful in this setting:
I find recommendations 41 and 43 particularly salient here, though I suspect the answer for most is dependent on this piece of info:

A zentensivist floats a swan
We elected to place a pulmonary artery catheter (and axillary art line) which yielded the following results:
- RAP 10
- RV 41/9
- PA 40/30 (33)
- PCWP 20
- MvO2 59
- Thermodilution cardiac index 1.7 (Fick CI 2.1)
- PvaCO2 10
This patient’s physical exam supported the diagnosis of insufficient cardiac output, which is confirmed based on these findings. That said, the clinical history and lung US were suggestive of pneumonia and secondary septic shock. In the end, this was felt to be mixed shock. Vasopressin was already discontinued, and the decision was made to initiate dobutamine on top of norepinephrine.
One may wonder, why not just trial this empirically?
Perhaps a reasonable option, though starting dobutamine caused the norepinephrine dose to double, despite increasing the measured cardiac output. Without the cardiac output information, it would have been difficult to interpret this response. (Anecdotally, I have noticed that patients with septic shock and coincident cardiac dysfunction sometimes do not tolerate the vasodilatory effects of the dobutamine).
Resolution
We switched from dobutamine to epinephrine and were able to wean norepi within the next 12 hours. Cardiac index had gradually improved from 1.7 to 2.9 over the course.
With such rapid resolution of cardiac dysfunction, I suspected the etiology proposed here by Glenn Hernandez:
Neuromuscular blockade, epoprostenol, all sedation, and the PA catheter were all withdrawn within 24 hours of presentation. Vasoactives were weaned within the following 12 hours. The patient was successfully extubated a few days later.
Crescendo-decrescendo critical care
When diagnostic uncertainty and illness severity are both high, it makes sense to pursue more invasive means to point the care path in the right direction.
I’m no fan of exhaustive work-ups, but I have the highest urgency about establishing the primary diagnoses by whatever means are necessary and prudent.
As Bob Lee put it:

As always, just because we started off invasively, doesn’t mean we needed to stay there. We removed nearly all the lines/tubes within 24-48 hours, and continued the minimally-invasive, maximally attentive critical care we’re used to.
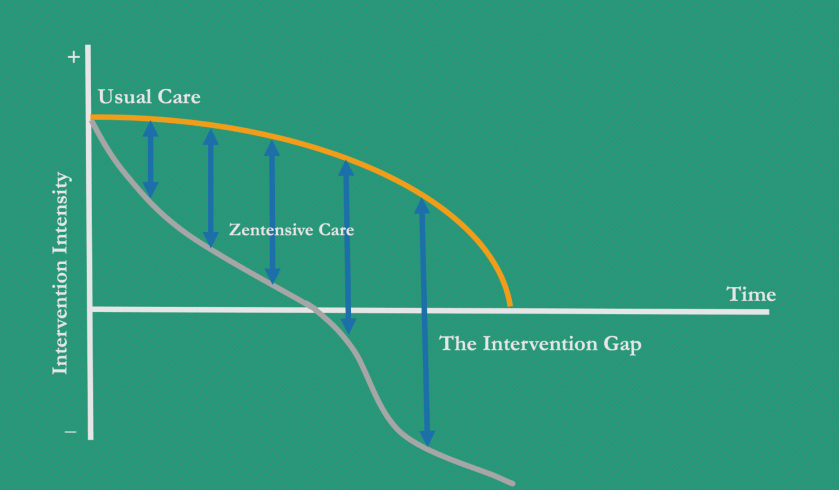
Unselected invasive devices probably don’t help anyone, and that’s probably how the PA catheter fell out of favor in the first place. I might have attempted this management plan with a standard central line to start, but with such high severity of illness, and the likelihood I’d remove it soon, the risk:benefit ratio seemed favorable for the PAC.
When organ failures and mortality are on the line, making the correct primary diagnosis is key. As soon as we’ve righted the ship, we can gracefully get out of the body’s way.
-MTS
Notes:
- Image by PublicDomainPictures from Pixabay



